Febuxostat
Atenurix
Dosage Availability (per box of 30’s):
20mg Film Coated Tablet
40mg Film Coated Tablet
80mg Film Coated Tablet
ANTI-HYPERURICEMIA


Dosage Availability (per box of 30’s):
20mg Film Coated Tablet
40mg Film Coated Tablet
80mg Film Coated Tablet
ANTI-HYPERURICEMIA
Each film coated tablet contains :
Febuxostat 20mg
Febuxostat 40mg
Febuxostat 80mg
Treatment of chronic hyperuricemia in conditions where urate deposition has already occurred (including a history, or presence of, tophus and/or gouty arthritis).
Hypersensitivity to Febuxostat or to any components of this product. Pregnancy and breastfeeding.
Adults
The recommended oral dose of FEBUXOSTAT is 20 mg or 40mg once daily with or without food, depending on the baseline serum uric acid (SUA) levels (20mg if suA < 7.5 mg/dL; 40mg if suA > 8.0 mg/dL). If serum uric acid is > 6 mg/dL (357 µmol/l) after 2-4 weeks, the dosage may be increased to 120 mg once daily. FEBUXOSTAT works sufficiently quickly to allow retesting of the serum uric acid after 2 weeks. The therapeutic target is to decrease and maintain serum uric acid below 6 mg/dl (357μmol/l). Gout flare prophylaxis of at least 6 months is recommended. Or as prescribed by the physician.
Elderly
No dose adjustment is required in the elderly.
Children (<18 years)
As there has been no experience in children and adolescents, the use of FEBUXOSTAT in such patients is not recommended.
Renal Impairment
No dosage adjustment is necessary in patients with mild or moderate renal impairment. The efficacy and safety have not been fully evaluated in patients with severe renal impairment (creatinine clearance <30 ml/min).
Hepatic Impairment
The recommended dosage in patients with mild hepatic impairment is 80 mg. Limited information is available in patients with moderate hepatic impairment. The efficacy and safety of FEBUXOSTAT has not been studied in patients with severe hepatic impairment (Child Pugh Class C).
Uric acid is the end product of purine metabolism in humans and is generated in the cascade of hypoxanthine > xanthine > uric acid. Both steps in the above transformations are catalyzed by xanthine oxidase. FEBUXOSTAT is a 2-arylthiazole derivative that achieves its therapeutic effect of decreasing serum uric acid by selectively inhibiting xanthine oxidase. FEBUXOSTAT is a potent, non-purine selective inhibitor of xanthine oxidase (NP-SIXO) with an in vitro inhibition Ki value less than one nanomolar. FEBUXOSTAT has been shown to potently inhibit both the oxidized and reduced forms of xanthine oxidase. At therapeutic concentrations FEBUXOSTAT does not inhibit other enzymes involved in purine or pyrimidine metabolism, namely, guanine deaminase, hypoxanthine guanine phosphoribosyltransferase, orotate phosphoribosyltransferase, orotidine monophosphate decarboxylase or purine nucleoside phosphorylase.
Absorption
FEBUXOSTAT is rapidly (tmax of 1.0-1.5 h) and well absorbed (at least 84%). After single or multiple oral 80 and 120 mg once daily doses, Cmax is approximately 2.8-3.2 µg/ml, and 5.0-5.3 µg/ml, respectively. Absolute bioavailability of the FEBUXOSTAT tablet formulation has not been studied. Following multiple oral 80 mg once daily doses or a single 120 mg dose with a high fat meal, there was a 49% and 38% decrease in Cmax and an 18% and 16% decrease in AUC, respectively. However, no clinically significant change in the percent decrease in serum uric acid concentration was observed where tested (80 mg multiple dose). Thus, FEBUXOSTAT may be taken without regard to food.
Distribution
The apparent steady state volume of distribution (Vss/F) of FEBUXOSTAT ranges from 29 to 75 l after oral doses of 10-300 mg. The plasma protein binding of FEBUXOSTAT is approximately 99.2%, (primarily to albumin), and is constant over the concentration range achieved with 80 and 120 mg doses. Plasma protein binding of the active metabolites ranges from about 82% to 91%.
Metabolism
FEBUXOSTAT is extensively metabolized by conjugation via uridine diphosphate glucuronosyltransferase (UDPGT) enzyme system and oxidation via the cytochrome P450 (CYP) system. Four pharmacologically active hydroxyl metabolites have been identified, of which three occur in plasma of humans. In vitro studies with human liver microsomes showed that those oxidative metabolites were formed primarily by CYP1A1, CYP1A2, CYP2C8 or CYP2C9 and FEBUXOSTAT glucuronide was formed mainly by UGT 1A1, 1A8, and 1A9
Elimination
FEBUXOSTAT is eliminated by both hepatic and renal pathways. Following an 80 mg oral dose of 14C-labeled FEBUXOSTAT, approximately 49% of the dose was recovered in the urine as unchanged FEBUXOSTAT (3%), the acyl glucuronide of the active substance (30%), its known oxidative metabolites and their conjugates (13%), and other unknown metabolites (3%). In addition to the urinary excretion, approximately 45% of the dose was recovered in the feces as the unchanged FEBUXOSTAT (12%), the acyl glucuronide of the active substance (1%), its known oxidative metabolites and their conjugates (25%), and other unknown metabolites (7%). FEBUXOSTAT has an apparent mean terminal elimination half-life (t1/2) of approximately 5 to 8 hours.
Patients with renal impairment
Following multiple doses of 80 mg of FEBUXOSTAT in patients with mild, moderate or severe renal insufficiency, the Cmax of FEBUXOSTAT did not change, relative to subjects with normal renal function. The mean total AUC of febuxostat increased by approximately 1.8-fold from 7.5 µg.h/mL in the normal renal function group to 13.2 µg.h/mL in the severe renal dysfunction group. The Cmax and AUC of active metabolites increased up to 2- and 4-fold, respectively. However, no dose adjustment is necessary in patients with mild or moderate renal impairment.
Patients with hepatic impairment
Following multiple doses of 80 mg of FEBUXOSTAT in patients with mild (Child-Pugh Class A) or moderate (Child-Pugh Class B) hepatic impairment, the Cmax and AUC of FEBUXOSTAT and its metabolites did not change significantly compared to subjects with normal hepatic function. No studies have been conducted in patients with severe hepatic impairment (Child- Pugh Class C).
Age
There were no significant changes observed in AUC of FEBUXOSTAT or its metabolites following multiple oral doses of FEBUXOSTAT in elderly as compared to younger healthy subjects.
Cardio-vascular disorders
Treatment with FEBUXOSTAT in patients with ischaemic heart disease or congestive heart failure is not recommended.
Acute gouty attacks (gout flare)
Febuxostat treatment should not be started until an acute attack of gout has completely subsided. As with other urate lowering medicinal products, gout flares may occur during initiation of treatment due to changing serum uric acid levels resulting in mobilization of urate from tissue deposits. At treatment initiation with FEBUXOSTAT flare prophylaxis for at least 6 months with an NSAID or colchicine is recommended. If a gout flare occurs during FEBUXOSTAT treatment, it should not be discontinued. The gout flare should be managed concurrently as appropriate for the individual patient. Continuous treatment with FEBUXOSTAT decreases frequency and intensity of gout flares.
Xanthine deposition
As with other urate lowering medicinal products, in patients in whom the rate of urate formation is greatly increased (e.g. malignant disease and its treatment, Lesch-Nyhan syndrome) the absolute concentration of xanthine in urine could, in rare cases, rise sufficiently to allow deposition in the urinary tract. As there has been no experience with FEBUXOSTAT, its use in these populations is not recommended.
Mercaptopurine/Azathioprine
FEBUXOSTAT use is not recommended in patients concomitantly treated with mercaptopurine/azathioprine.
Theophylline
FEBUXOSTAT should be used with caution in patients concomitantly treated with theophylline and theophylline levels should be monitored in patients starting FEBUXOSTAT therapy.
Liver disorders
During the phase 3 clinical studies, mild liver function test abnormalities were observed in patients treated with FEBUXOSTAT (3.5%). Liver function test is recommended prior to the initiation of therapy with FEBUXOSTAT and periodically thereafter based on clinical judgement.
Thyroid disorders
Increased TSH values (>5.5 µIU/ml) were observed in patients on long-term treatment with FEBUXOSTAT (5.0%) in the long term open label extension studies. Caution is required when FEBUXOSTAT is used in patients with alteration of thyroid function.
DRUG INTERACTIONS
Mercaptopurine/Azathioprine
Although interaction studies with FEBUXOSTAT have not been performed, inhibition of xanthine oxidase (XO) is known to result in an increase in mercaptopurine or azathioprine levels. On the basis of the mechanism of action of FEBUXOSTAT on XO inhibition concomitant use is not recommended. Drug interaction studies of FEBUXOSTAT with cytotoxic chemotherapy have not been conducted. No data is available regarding the safety of FEBUXOSTAT during cytotoxic therapy.
Theophylline
Although interaction studies have not been performed with FEBUXOSTAT, inhibition of XO may cause an increase in the theophylline level (inhibition of the metabolism of theophylline has been reported with other XO inhibitors). Hence caution is advised if these active substances are given concomitantly, and theophylline levels should be monitored in patients starting FEBUXOSTAT therapy.
Naproxen and other inhibitors of glucuronidation
FEBUXOSTAT metabolism depends on UGT enzymes. Medicinal products that inhibit glucuronidation, such as NSAIDs and probenecid, could in theory affect the elimination of FEBUXOSTAT. In healthy subjects concomitant use of FEBUXOSTAT and naproxen 250mg BID was associated with an increase in FEBUXOSTAT exposure (Cmax 28%, AUC 41% and t1/2 26%). In clinical studies the use of naproxen or other NSAIDs/Cox-2 inhibitors was not related to any clinically significant increase in adverse events. FEBUXOSTAT can be co-administered with naproxen with no dose adjustment of FEBUXOSTAT or naproxen being necessary.
Inducers of glucuronidation
Potent inducers of UGT enzymes might possibly lead to increased metabolism and decreased efficacy of FEBUXOSTAT. Monitoring of serum uric acid is therefore recommended 1-2 weeks after start of treatment with a potent inducer of glucuronidation. Conversely, cessation of treatment of an inducer might lead to increased plasma levels of FEBUXOSTAT.
Colchicine/Indomethacin/Hydrochlorothiazide/Warfarin
FEBUXOSTAT can be co-administered with colchicine or indomethacin with no dose adjustment of FEBUXOSTAT or the co-administered active substance being necessary.
No dose adjustment is necessary for FEBUXOSTAT when administered with hydrochlorothiazide. No dose adjustment is necessary for warfarin when administered with FEBUXOSTAT. After initiation of FEBUXOSTAT therapy, monitoring of anticoagulant activity should be considered in patients receiving warfarin or similar agents.
Desipramine/CYP2D6 substrates.
FEBUXOSTAT was shown to be a weak inhibitor of CYP2D6 in vitro. In a study in healthy subjects, 120 mg FEBUXOSTAT resulted in a mean 22% increase in AUC of desipramine, a CYP2D6 substrate indicating a potential weak inhibitory effect of FEBUXOSTAT on the CYP2D6 enzyme in vivo. Thus, co-administration of FEBUXOSTAT with other CYP2D6 substrates is not expected to require any dose adjustment for those compounds.
Antacids
Concomitant ingestion of an antacid containing magnesium hydroxide and aluminum hydroxide has been shown to delay absorption of FEBUXOSTAT (approximately 1 hour) and to cause a 32% decrease in Cmax, but no significant change in AUC was observed. Therefore, FEBUXOSTAT may be taken without regard to antacid use.
A total of 2531 subjects received at least one dose of FEBUXOSTAT (10 mg – 300 mg) in clinical studies.
Phase 3 randomized controlled studies
In randomized controlled phase 3 clinical studies, >1,000 patients have been treated with the recommended doses of 80 mg or 120 mg (536 subjects enrolled in a 28 week study and 507 subjects enrolled in a 52 weeks study). The treatment-related events (ADRs) were mostly mild or moderate in severity. The most commonly reported ADRs (investigator assessment) are liver function abnormalities (3.5%), diarrhea (2.7%), headache (1.8%), nausea (1.7%), and rash (1.5 %). A numerically greater incidence of investigator-reported cardiovascular events was observed in the FEBUXOSTAT total group compared to the allopurinol group in the pivotal Phase III (1.3 vs 0.3 events per 100 PYs) and long-term extension studies (1.4 vs 0.7 events per 100 PYs), although no statistically significant differences were found and no causal relationship with FEBUXOSTAT was established. Identified risk factors among these patients were a medical history of atherosclerotic disease and/or myocardial infarction, or of congestive heart failure.
Common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100) and rare (≥ 1/10,000 to < 1/1000) adverse reactions suspected (investigator assessment) to be drug related occurring in the 80 mg/120 mg treatment groups and reported more than once in the total FEBUXOSTAT treatment group are as follows; Investigations: Uncommon; blood amylase increase, platelet count decrease, blood creatinine increase, hemoglobin decrease, blood urea increase, LDH increase, triglycerides increase. Cardiac Disorders: Rare; palpitations. Gastrointestinal Disorders: Common; diarrhea*, nausea* Nervous System Disorders: Common; headache. Uncommon; dizziness, paraesthesia, somnolence, altered taste. Renal and Urinary Disorders: Uncommon; Nephrolithiasis, hematuria, pollakiuria. Rare; renal insufficiency. Skin and Subcutaneous Tissue Disorders: Common; rash**. Uncommon; dermatitis, urticaria, pruritis. Musculoskeletal and Connective Tissue Disorders: Uncommon; Arthralgia, arthritis, myalgia, muscle cramp, musculoskeletal pain. Metabolism and Nutrition Disorders: Uncommon; weight increase, increased appetite. Vascular Disorders: Uncommon; hypertension, flushing, hot flush. General Disorders and Administration Site Conditions: Uncommon; fatigue, edema, influenza like symptoms. Rare: asthenia, thirst. Hepato-Biliary Disorders: Common; LFT abnormalities. Psychiatric Disorders: Uncommon; libido decreased. Rare: nervousness, insomnia. * Diarrhea, nausea and vomiting are more frequent in patients concomitantly treated with colchicine ** No serious rashes or severe hypersensitivity reactions were noted in the clinical studies.
Long-term open label extension studies – In the long-term open label extension studies, the number of patients treated with FEBUXOSTAT 80 mg/120 mg up to 1 year was 906, up to 2 years was 322, up to 3 years was 57, and up 4 years was 53. The treatment-related events reported during the long-term extension studies were similar to those reported in the Phase 3 studies. The most commonly reported treatment-related events (investigator assessment) are: liver function abnormalities, diarrhea, headache, rash, and hypertension. The following treatment-related events were reported more than once in the total FEBUXOSTAT treatment group and were reported as uncommon in subjects taking FEBUXOSTAT 80 mg/120 mg in long-term extension studies (up to 4 years, >1,900 Patient-years of exposure). These treatment-related events were either not reported or reported at a lower frequency for these doses, in the pivotal Phase 3 studies: Diabetes, hyperlipidemia, insomnia, hypoaesthesia, ECG abnormal, cough, dyspnea, skin discoloration, skin lesion, bursitis, proteinuria, renal insufficiency, erectile dysfunction, blood potassium increase, blood TSH increase, lymphocyte count decreased, WBC decrease.
Pregnancy
Data on a very limited number of exposed pregnancies have not indicated any adverse effects of FEBUXOSTAT on pregnancy or on the health of the fetus/new born child. Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryonal/fetal development or parturition. The potential risk for human is unknown. FEBUXOSTAT should not be used during pregnancy.
Breast feeding
It is unknown whether FEBUXOSTAT is excreted in human breast milk. Animal studies have shown excretion of this active substance in breast milk and an impaired development of suckling pups. A risk to a suckling infant cannot be excluded. FEBUXOSTAT should not be used while breast-feeding.
Foods, Drugs, Devices and Cosmetics Act prohibits dispensing without a prescription.
STORE AT TEMPERATURES NOT EXCEEDING 30OC
Febuxostat (Atenurix®) 20mg Film Coated Tablet X 30 tablets / box / Alu-alu blister pack
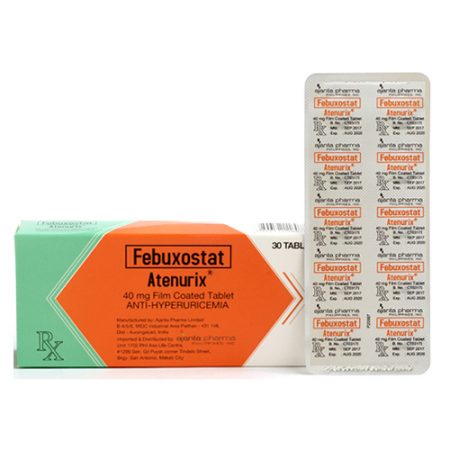
Febuxostat (Atenurix®) 40mg Film Coated Tablet X 30 tablets / box / Alu-alu blister pack
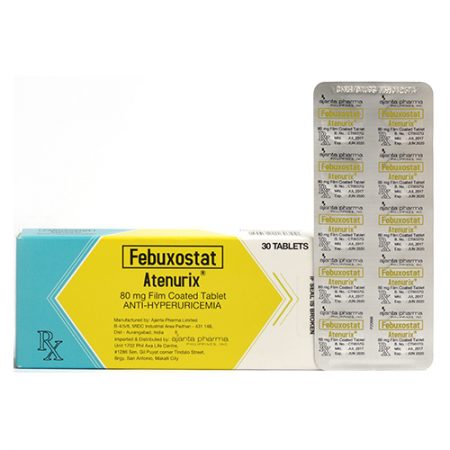
Febuxostat (Atenurix®) 80mg Film Coated Tablet X 30 tablets / box / Alu-alu blister pack
Atenurix® is a registered trademark of Ajanta Pharma Philippines, Inc.


ABOUT US
A wholly owned subsidiary of a global pharmaceutical supplier
OUR PRODUCTS
First to market pharmaceutical compounds, unique therapeutically optimized combinations, value branded equivalents
CAREERS @ AJANTA
Looking for a challenging and rewarding career?
Apply here
PATIENT RESOURCES
Links to online educational materials